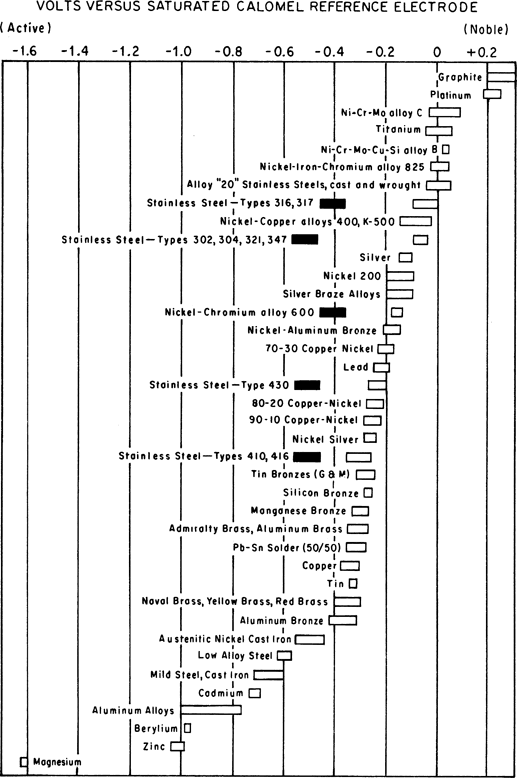
The galvanic series (or electropotential series) determines how well two metals will work together to make a battery. When two metals are submerged in an electrolyte, while electrically connected, the less noble (base) will experience galvanic corrosion. The rate of corrosion is determined by the electrolyte and the difference in nobility. The difference can be measured as a difference in voltage potential. Galvanic reaction is the principle upon which batteries are based.

The noble metals are a group of metals that resist oxidation and corrosion in moist air. They are the opposite of the base metals, which more readily oxidize and corrode. Electrolytes are liquid substances that act as a medium to conduct electricity and oxidizes and corrodes the metals. Different metals and electrolytes product different strength of batteries. An electrolyte is full of ions, which are atoms that have some sort of net electric charge, either positive or negative. Batteries employ an electrolyte of some sort, both to conduct electricity between the metals in batteries and to store energy on the metals themselves. The type used in a battery depends on the battery’s type and purpose. Most car batteries, for example, use one that contains sulfuric acid, which is why they require careful handling. Alkaline batteries use an alkaline solution. Lithium batteries use a special organic electrolyte that freezes at much lower temperatures than more traditional water-based ones. Carbon batteries use zinc chloride and ammonium chloride as their electrolyte. All kinds of things can be an electrolyte including vinegar, lye, lemon juice, bleach and even ocean salt water can be an electrolyte.
The following is the galvanic series for an electrolyte of stagnant (that is, low oxygen content) seawater.
The order may change in different environments.
-
- Graphite
- Palladium
- Platinum
- Gold
- Silver
- Titanium
- Stainless steel 316 (passive)
- Stainless Steel 304 (passive)
- Silicon bronze
- Stainless Steel 316 (active)
- Monel 400
- Phosphor bronze
- Admiralty brass
- Cupronickel
- Molybdenum
- Red brass
- Brass plating
- Yellow brass
- Naval brass 464
- Uranium 8% Mo
- Niobium 1% Zr
- Tungsten
- Tin
- Lead
- Stainless Steel 304 (active)
- Tantalum
- Chromium plating
- Nickel (passive)
- Copper
- Nickel (active)
- Cast iron
- Steel
- Indium
- Aluminum
- Uranium (pure)
- Cadmium
- Beryllium
- Zinc plating (see galvanization)
- Magnesium